Locate sample inventory
Track and manage your sample inventory in one centralized database. LIMSense also lets you create views of sample storage so you can search for exact locations.

Click image to enlarge

Click image to enlarge
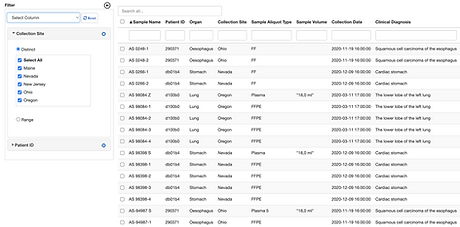
Fast multi-criteria search
Perform multi-criteria searches on the clinical, patient or sample meta data parameters you create to quickly find samples that meet specific criteria.
“At Singular Bio we surveyed a number of sample tracking and management systems to map onto our assay workflow and we were sold on the flexibility and ease of use of the AduroSys LIMSense platform. The final product fully met our needs and delighted our lab team who found it very intuitive and easy to use.”
- Singular Bio
See how LIMSense can work in your lab.
Click image to enlarge
Easily consolidate data
User-defined sample collection templates let you consolidate multiple data sets from different collection sites easily. Map only the parameters you want to import, and create standardized vocabularies to consolidate specific parameters across data sets.
Eliminate duplicates
Just create a set of parameters that map to your spreadsheet. LIMSense guides you through data upload and verification. Duplicates are identified automatically to let you decide what actions to take before import. Any issues with controlled vocabulary are auto-corrected or flagged for review.

Click image to enlarge
Easy Implementation and Integration
Transitioning to a LIMS or integrating a new LIMS into your lab workflow doesn’t have to be overwhelming. The AduroSys implementation team has decades of LIMS experience, and provides you with full support throughout the process to ensure an easy transition.

Planning. We meet with your team to understand your immediate and long-term goals and lab workflow.

Design. We set up your LIMS, create your workflows and quick access to features you need. But you have access to all features, all the time.

Implementation. We train your team and provide support through implementation in the lab so you’re up and running quickly.

Support. Whenever you have questions or need to update your workflows, we’re ready to assist.
LIMSense Features
Sample & Experiment Tracking
Lab Integration
-
Sample status tracking (requests, registration, shipments, and destruction)
-
Sample lineage, aliquoting and derivatives tracking
-
Sample batch processing
-
Sample inventory management
-
Sample tracking in and out of storage locations
-
Full chain of custody
-
Configurable sample management workflows
-
User-defined meta data
-
Data import via template mapping
-
Reporting with charts and dashboard
-
Multi-criteria search
-
Integration with external systems
-
Full Application Programming Interface (API) support
-
Cloud-hosted deployments

Easy on your budget
LIMSense gives labs all the functionality they need and will actually utilize. A simple, flat-fee subscription model gives you access to every feature. It’s more cost effective than larger, more complex multi-application systems.

Easy transition from spreadsheets
LIMSense eliminates manual spreadsheet manipulation, and makes it much easier to maintain data for a continually expanding sample inventory. Simple to use features and an intuitive interface take the stress out of the transition. Anyone can start using it right away.
Plate Tracking
-
Manage high-throughput plate processing
-
Instrument worklist generation
-
Auto-generate plate maps
-
User defined plate dimension
-
Heatmap visualization of plate data
Security & Compliance
-
Role-based security
-
Audit trail
-
21 CFR Part 11-compliant application
